My Car Battery Has White Or Blue Powder – Why Does It Happen And How Can It Be Fixed?
Have you noticed a white or blue powder around the battery terminals? You must be wondering what causes the powder formation. And whether it negatively impacts your battery performance or not.
So, why does my car battery have white or blue powder? The white powder is an end product of an electrochemical reaction between copper connectors and the lead terminals. This results in a white anhydrous copper sulfate or lead sulfate. On exposure to moisture, the white powder turns blue when hydrated.
This article reviews the common causes of white or blue powder on the terminals and ways of removing it. In addition, it also addresses key ways of preventing the formation of the powder.

Car Battery Has White Or Blue Powder: What is It?
The white powder forms first as anhydrous copper sulfate or lead sulfate. On exposure to environmental moisture, the white anhydrous copper sulfate turns into a blue hydrated copper sulfate.
On the other hand, blue powder may form if the battery is exposed to a moist environment. Thus, the chemical reaction is accelerated by the presence of salts in the sulfuric acid leading to a fast response.

If not checked, the powder forms on the terminal affecting its power supply to the electrical system. And the first signs are seen with dimmed headlights.
Why Does Car Battery Have White Or Blue Powder? How to Control It?
Below are the causes of these electrochemical reactions on the battery terminals.
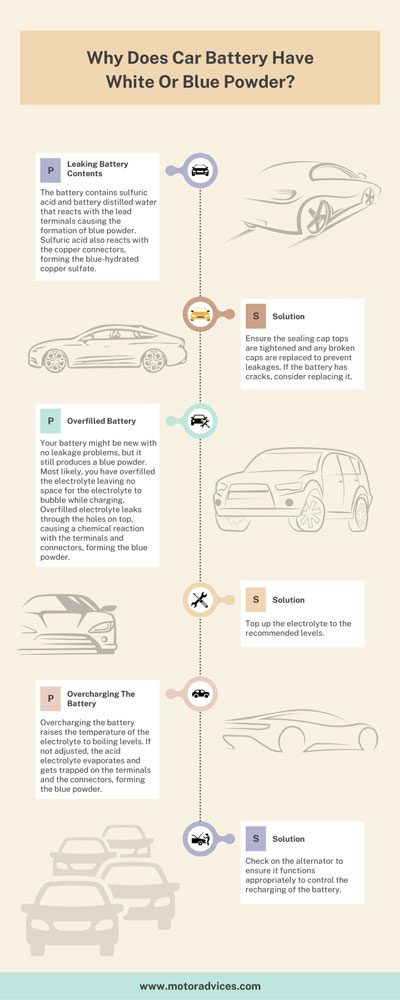
Leaking Battery Contents
The battery contains sulfuric acid and battery distilled water that reacts with the lead terminals causing the formation of blue powder. Sulfuric acid also reacts with the copper connectors, forming the blue-hydrated copper sulfate.

Solution
Ensure the sealing cap tops are tightened and any broken caps are replaced to prevent leakages. If the battery has cracks, consider replacing it.

Overfilled Battery
Your battery might be new with no leakage problems, but it still produces a blue powder. Most likely, you have overfilled the electrolyte leaving no space for the electrolyte to bubble while charging.
Overfilled electrolyte leaks through the holes on top, causing a chemical reaction with the terminals and connectors, forming the blue powder.

Solution
Top up the electrolyte to the recommended levels.
Overcharging The Battery
Overcharging the battery raises the temperature of the electrolyte to boiling levels. If not adjusted, the acid electrolyte evaporates and gets trapped on the terminals and the connectors, forming the blue powder.
Solution
Check on the alternator to ensure it functions appropriately to control the recharging of the battery.
How To Remove The Blue Powder From The Battery Terminals?

Accumulating the blue powder has negative impacts and needs to be removed without damaging the terminals. Below are some simple hacks on removing corrosion step by step.
Step 1: Prepare a baking soda and water mixture in a plastic container. Use two tablespoons of baking soda in 500mls of water.
Step 2: Disconnect the battery connectors starting with the negative connector. Use a screwdriver to loosen the connectors without damaging the lead terminals.
Step 3: Apply the baking soda mixture on the battery terminals and let it react for 5 minutes. Scrub off the powder using a brush. Check on the copper connectors and scrub any powder attached to them using the baking soda mixture.

Step 4: Rinse the remaining residuals with clean water and dry the terminals with a cloth. Apply petroleum jelly on the terminals to prevent further corrosion.
Step 5: Replace the connectors starting with the positive terminal connector and then the negative connector. Tighten the screws for a tight connection.
How To Prevent The Formation Of Blue Powder On The Battery Terminals?
You can perform the following maintenance practices to prevent your battery terminals from corroding.

1. Apply Anti-Corrosive Sprays
Spray anti-corrosive agents on the terminals once in a while to keep them from rusting.
2. Apply Dielectric Grease On The Terminals
Apply grease or petroleum jelly on the terminals to keep off moisture from attacking the battery terminals. Ensure you apply a thick layer on every part of the terminal.
3. Inspect The Battery Terminals Regularly
The battery keeps leaking the acidic contents as they get old. Inspect the battery regularly to clean the leaks and repair any problematic parts.

4. Do Not Overcharge Or Undercharge The Battery
Overcharging causes the acidic contents to leak and cause a chemical reaction with the terminal and the connectors. On the other hand, an undercharged battery causes corrosion on the negative terminal.
Various Colors Present On The Battery Terminals And What They Represent
Have you noticed different colors on your battery terminals? No need to worry; some of these color changes occur due to varying stages of chemical reactions.

Initially, you may only notice white powder and later find a blue powder on your battery terminals. Other times you may only see brown powder.
Below are the possible powder colors on your battery terminals and what they represent.
- Brown powder – The brown powder results from rust on the terminals, which is caused by moisture and oxygen on the terminals. When the lead terminals are exposed to oxygen and water, they oxidize to form a gray-brownish powder.
- White powder – The white powder forms as a product of the chemical reaction between the lead terminals and the sulfuric acid in the battery, forming lead sulfate.
It may also be due to the reaction between the copper connectors and the sulfuric acid forming anhydrous copper sulfate. Both products are white.
- Blue powder – Once the white powder forms on the terminals, it changes to blue powder due to exposure to moisture that further oxidizes it to a blue powder.
Effect Of The White/Bluish Powder On The Battery Terminals
The blue powder forms on the terminals inhibiting the normal functioning of the battery. Below are some of the negative impacts of the powder.

- The powder decreases the battery life by almost half its life expectancy. In extreme cases, the battery leaks sulfuric acid causing damage to other engine parts.
- Electrical short circuits – If not removed, the powder can accumulate and become a conducting substance that causes electrical short circuits through contact.
- Starting problems – the powder forms on the terminals and, at times, prevents the free flow of current to the vehicle’s electrical system.
- Poor recharging – the vehicle through the alternator ensures the battery keeps recharging as you drive. However, the blue powder hinders the free flow of electrons reducing the recharging efficiency.
FAQs
Here are the frequently asked questions to better understand the powder forming on the battery terminals.
Yes. The blue powder, if not removed, can cause starting problems, electrical shorts, and even decrease the battery’s lifespan.
The white powder results from a chemical reaction between the copper connectors and the sulfuric acid in the battery. This reaction produces a white powder known as anhydrous copper sulfate.
Sulfuric acid leaks when overcharging, causing the acid to leak and get in contact with the connectors.
Yes. The corrosion on the terminals is an electrochemical process that causes the loss of electrons. These electrons accumulate and interfere with the normal recharging process.
Over time, the battery stops recharging and loses its power through chemical reactions.
Conclusion
The blue powder on your battery terminals results from corrosion on the lead terminals. Thus, the sulfuric acid in the battery leaks to the lead terminals, causing a chemical reaction that forms the white anhydrous copper sulfate or lead sulfate.
Accordingly, the white powder is oxidized in moisture to the blue powder you often see on your terminals. To remove this blue powder, clean it with water and baking soda.

